The Organic Electronic Ion Pump (OEIP)
Flow-free delivery bio-substances at high spatiotemporal resolution (In vitro applications)
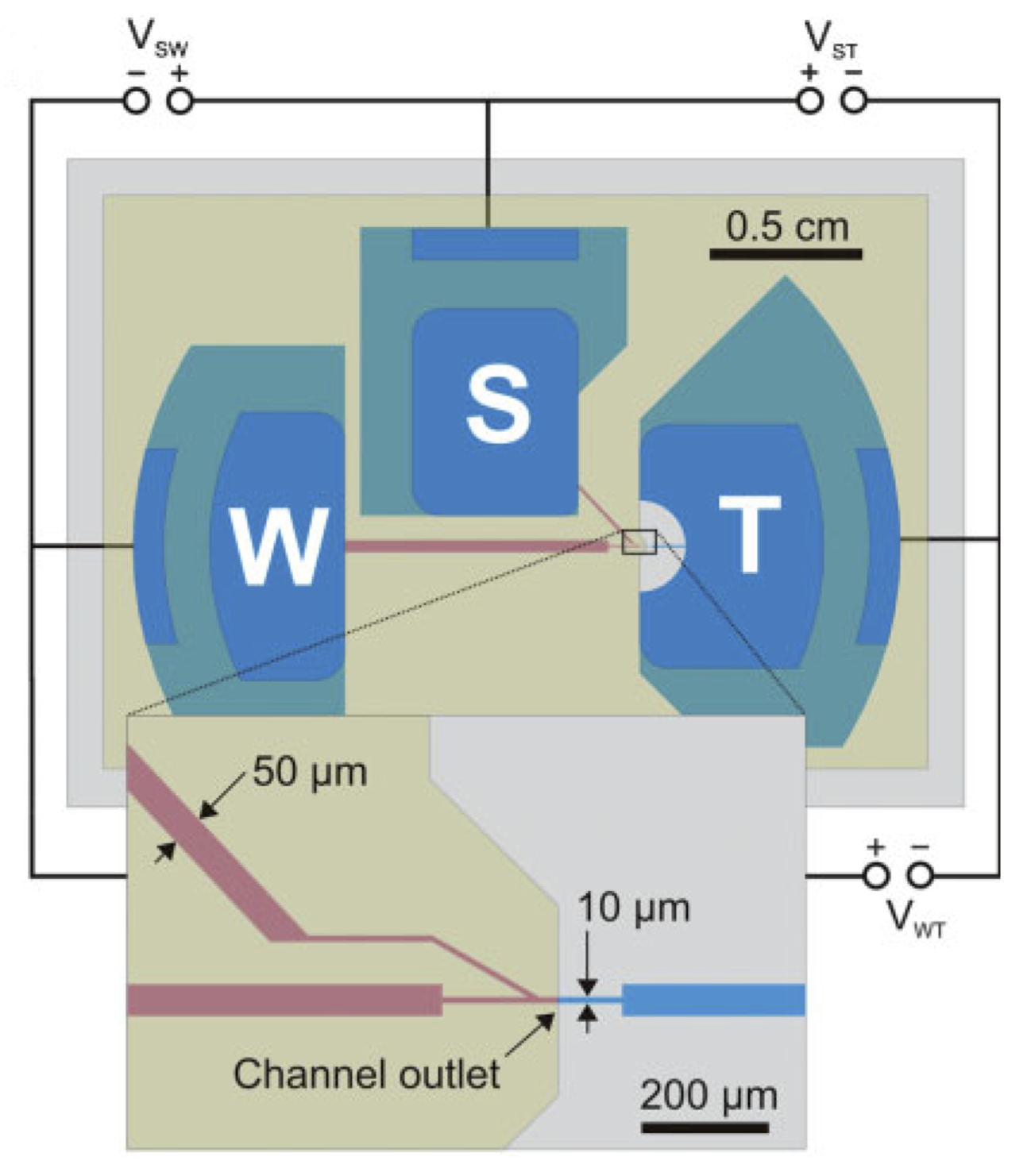
Miniaturized OEIPs are based on solid state polymers (plastics) that conduct both ions and electronic charges. They are manufactured using standard microfabrication techniques commonly used in electronics industry. The OEIPs translates electronic addressing signals into precise delivery of bio-substances at micrometer resolution and at high temporal resolution. OEIPs can easily be integrated into standard cell- and tissue growth plates, and can also be combined with various microscopy setups, incubators etc. OEIPs generate well-controlled, dynamic signal patterns at high spatiotemporal resolution far beyond what is possible using for instance micro-fluidics.
How does it work?
Cells are seeded within the target compartment (T) including suitable aqueous growth medium and desired bio-substance is dissolved in the source (S) electrolyte. By addressing the electrochemical electrodes, S, T and W, the substance is selectively transported, via electrophoresis, through the polyelectrolyte channel and is then delivered to cells at the outlet spot (see figure above).
Technical specifications
Time delay (from addressing to delivery) 1 sDelivery rate 1-100 *10-12 mole/s
Generated concentration at the target 0.1 nM to 100 mM
Size of the outlet 10 μm to 1 mm
Addressing voltages 0.1 to 20 V
Substances that can be delivered H+, K+, alkali ions, acetylcholine, glutamate, aspartate, GABA
Examples of applications
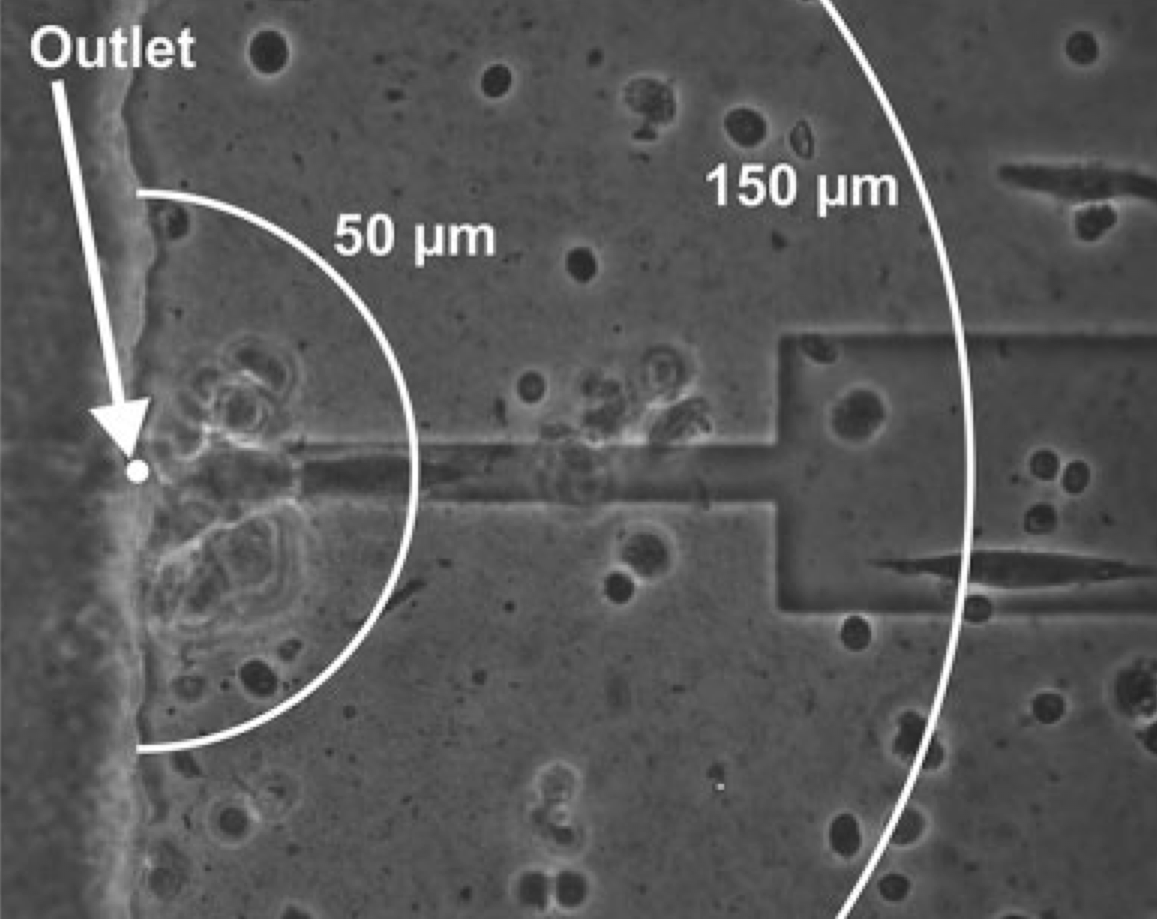
Delivering neurotransmitters at the micron scale is crucial in order to better understand the characteristics of signaling cascades in neurons, tissues etc. The OEIP, with an outlet of 10 micron (see the top figure to the right) was utilized to deliver acetylcholine (ACh) in close proximity to human SH-SY5Y neuroblastoma cells. By varying the time for which the addressing voltage is applied we control the amount of ACh delivered, which results in dynamic control of the intracellular signaling of the SH-SY5Y cells. In the case presented below to the right, the OEIP is inducing oscillation of the intracellular Ca2+ concentration meaning that the OEIP can artificially control this important and global signaling mechanism in the cell.
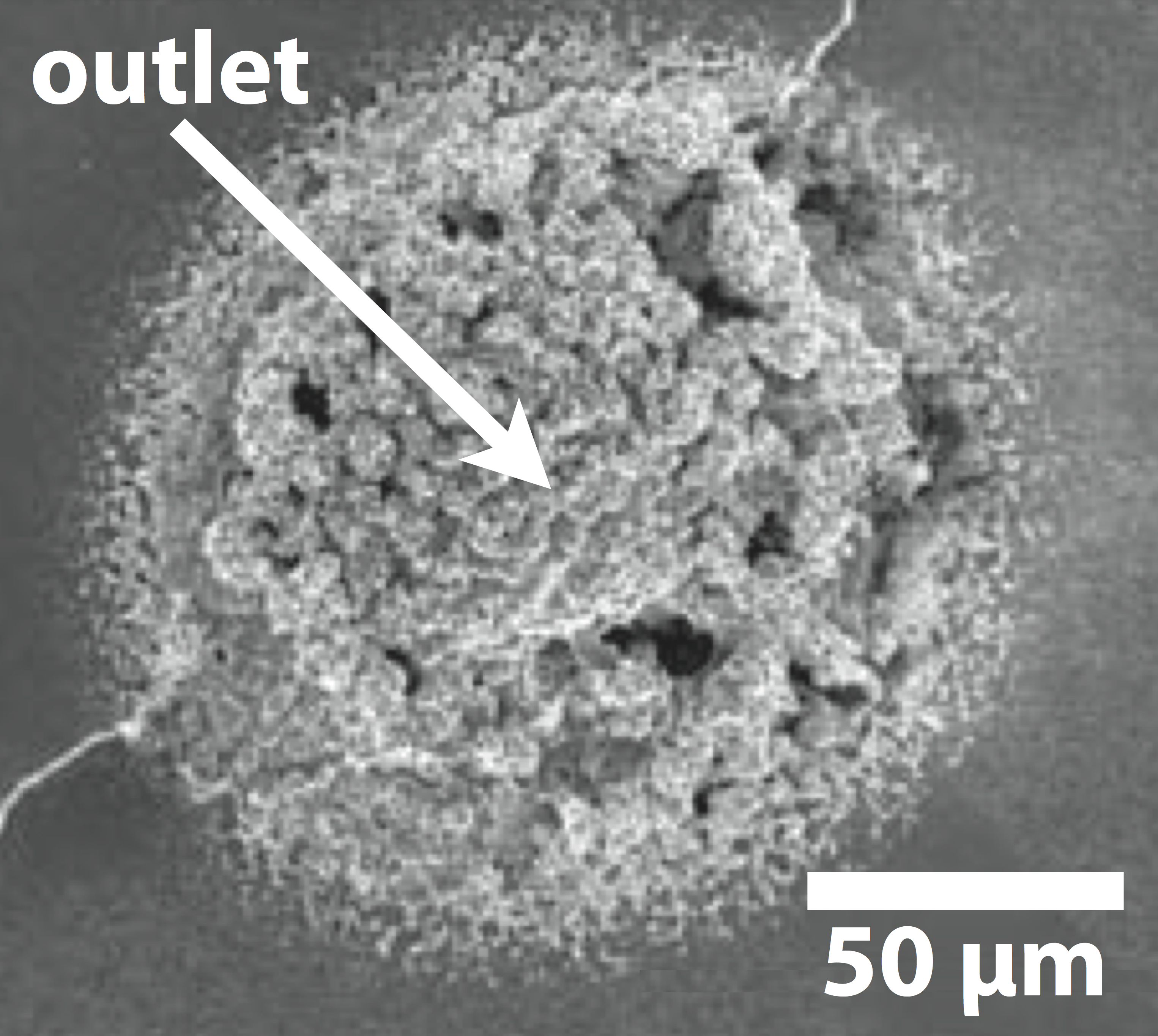
Amyloids are fibrous protein aggregates and are the origin to several neurodegenerative diseases, such as Parkinson’s disease. Various ionic substances are known to affect and control the formation and morphology of amyloids at the nano to micro scale. To better understand the characteristics and growth stages of amyloids it is important to deliver biochemical signals at flow-free conditions and at ultra-high resolution. Here, we have used the OEIP to electronically control the formation of amyloids. We found that small variations of OEIP ion delivery cause huge differences in the shape of the amyloids.
References
Flow-free delivery of bio-substances in vivo (In vivo applications)
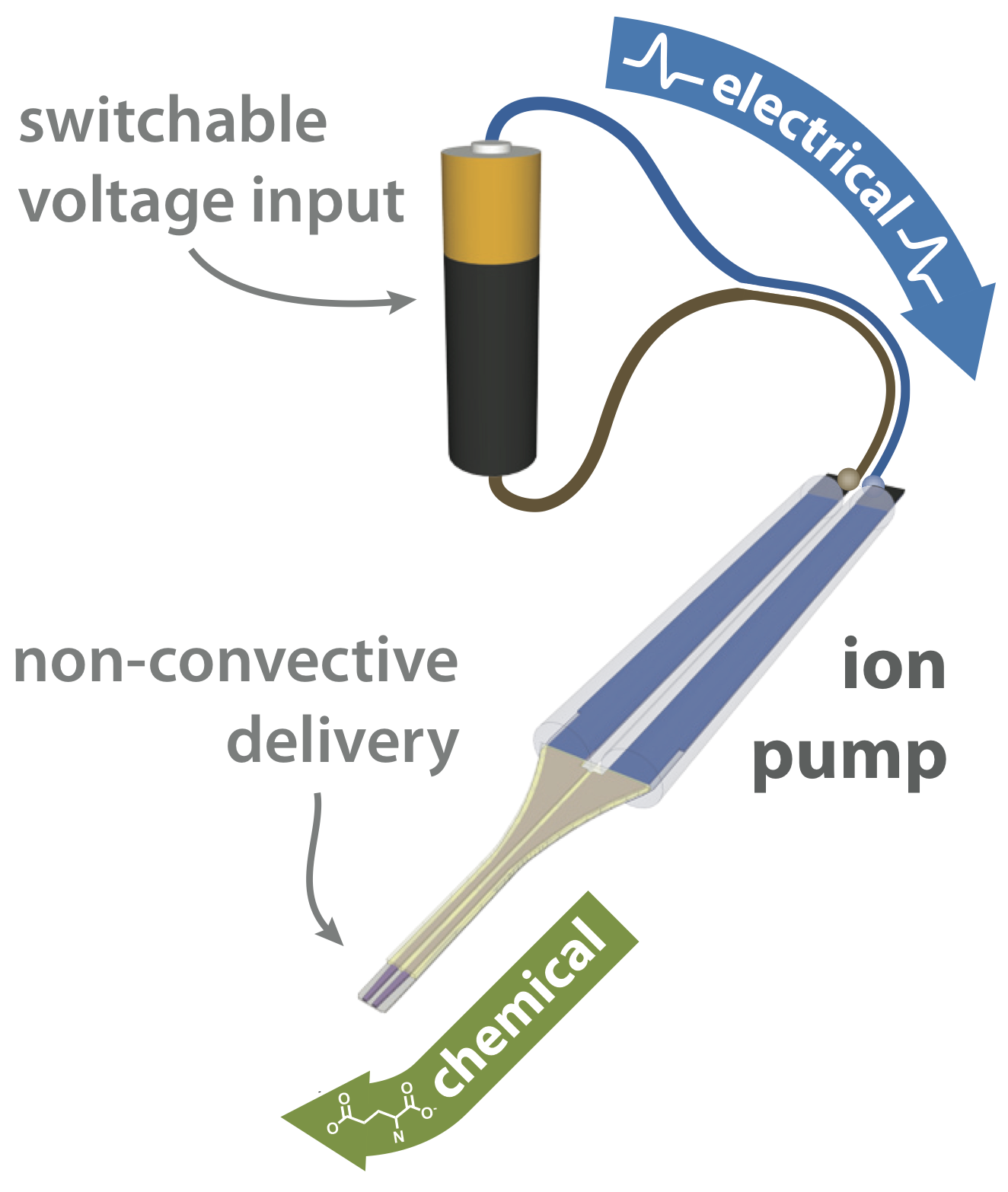
OEIPs have been adapted for use in an encapsulated, format for in vivo applications. As with the planar in vitro version, they can precisely translate electronic addressing signals into precise delivery of bioactive substances, without requiring fluid flow; only the substance is delivered, not the solution. In the in vivo adressing form, silicone chambers form electrolyte reservoirs, with the electrodes embedded within. The well-defined delivery tip can be placed in – or in contact with – tissue, adjacent to cells, or in close proximity to a region of three-dimensional space to which substances should be delivered. All components of the device are based on polymers, resulting in a pliable, biocompatible in vivo device.
How does it work?
The substance to be delivered is dissolved in the source electrolyte, and the delivery tip is placed into the target system. When voltage is applied, an electrochemical circuit is formed between the two reservoirs, connected by an ion conducting channel. As the substance being delivered is electrophoretically transported through the channel, it is transferred into the target system by rapid ion exchange through the exposed thin film at the delivery tip. Governed by strict electrochemical relationships, the electronic current measured in the control circuit is thus a direct indication of the “flow rate” of molecules into the target system.
Technical specifications
Time delay (addressing to delivery) As low as 1 sDelivery rate 1 — 100 pmol/s
Size at the delivery tip ~6 mm x ~ 1 mm
Size of the reservoir section ~30 mm x ~3 mm
Characterisitc substances that can be delivered H+, K+, alkali ions, acetylcholine, glutamate, aspartate, GABA
Examples of applications
3D in vitro delivery
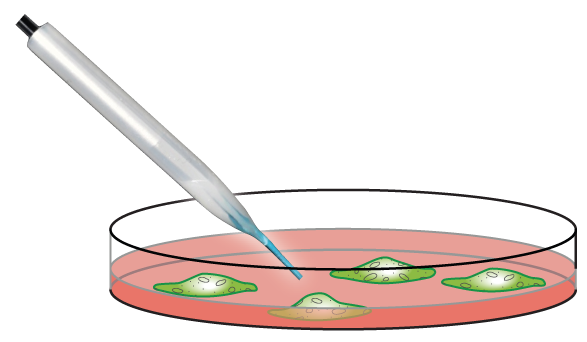
Three-dimensional positioning of delivery is possible by simply mounting the in vivo OEIP on a standard micromanipulator. Such a setup was used to demonstrate device efficacy through delivery of the neurotransmitter glutamate to cortical neurons.
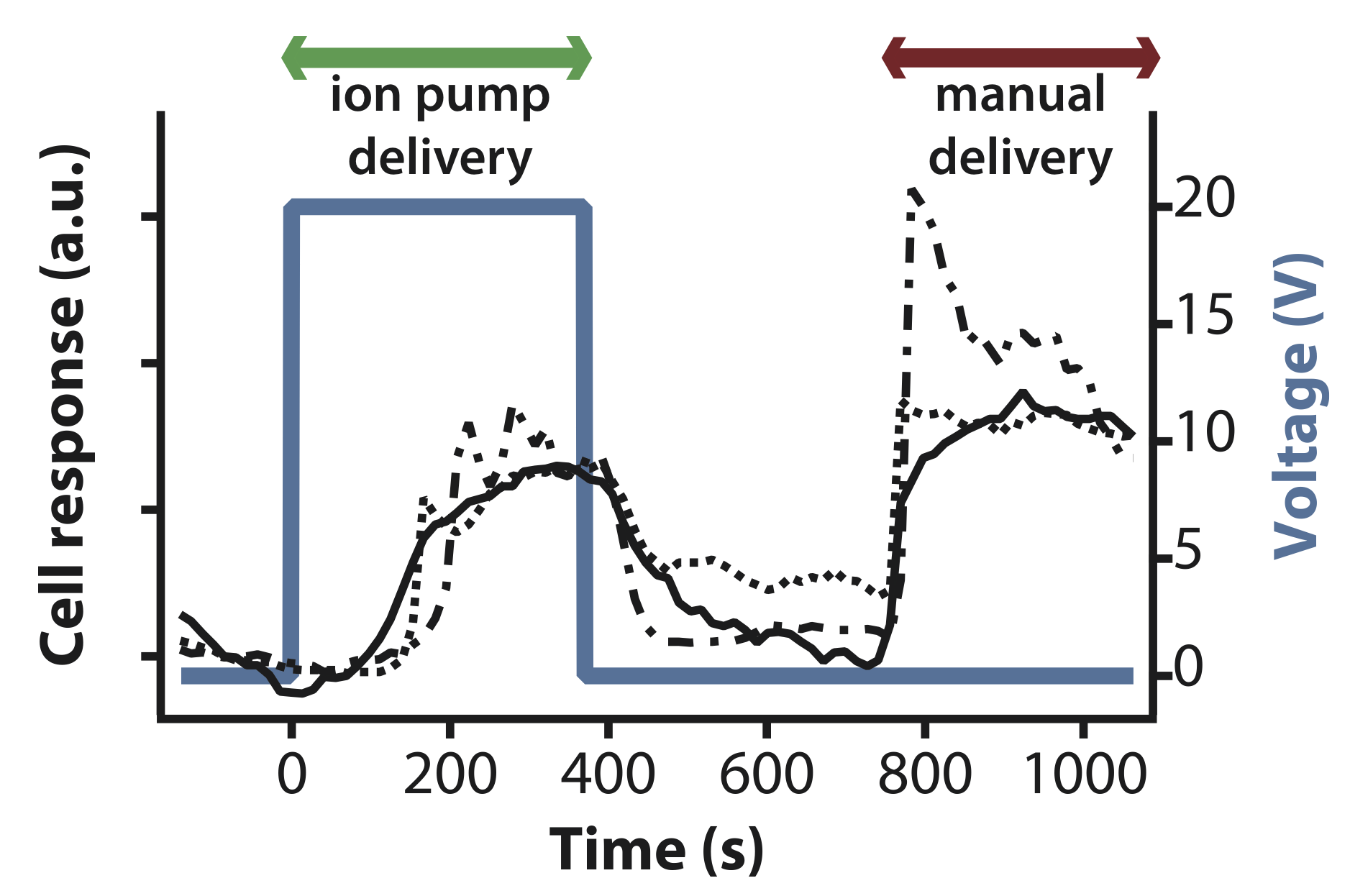
OEIP delivered glutamate elicited a similar physiological response to manually administered glutamate. With careful use of the micromanipulator, the device can be placed after cell culturing, enabling the practitioner to choose one particular region of cells for study.
3D in vivo delivery
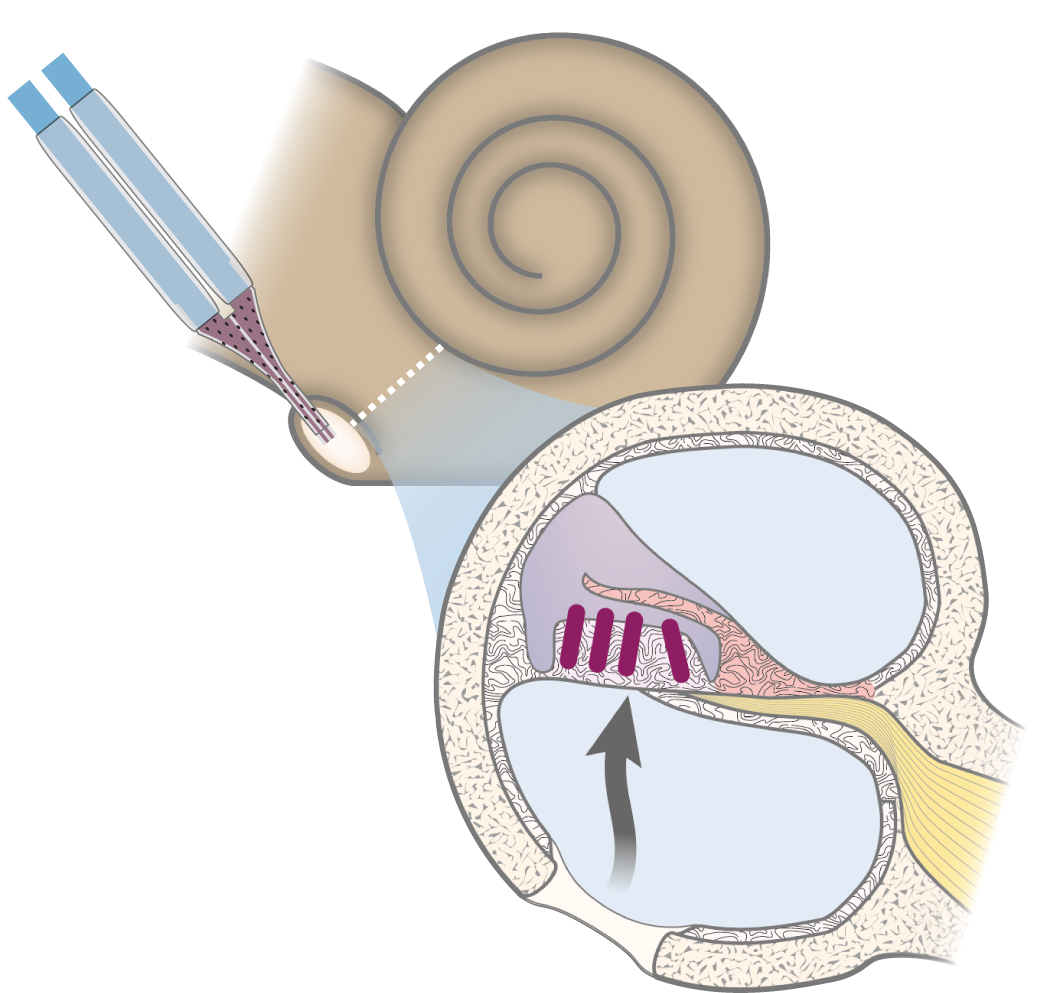
The cochlea is an ideal system for demonstrating efficacy in vivo: the two types of sound-transducing hair cells utilize different neurotransmitters. Glutamate can thus be delivered, and its effect observed on a specific type of cells (inner hair cells) within a heterogenous population.
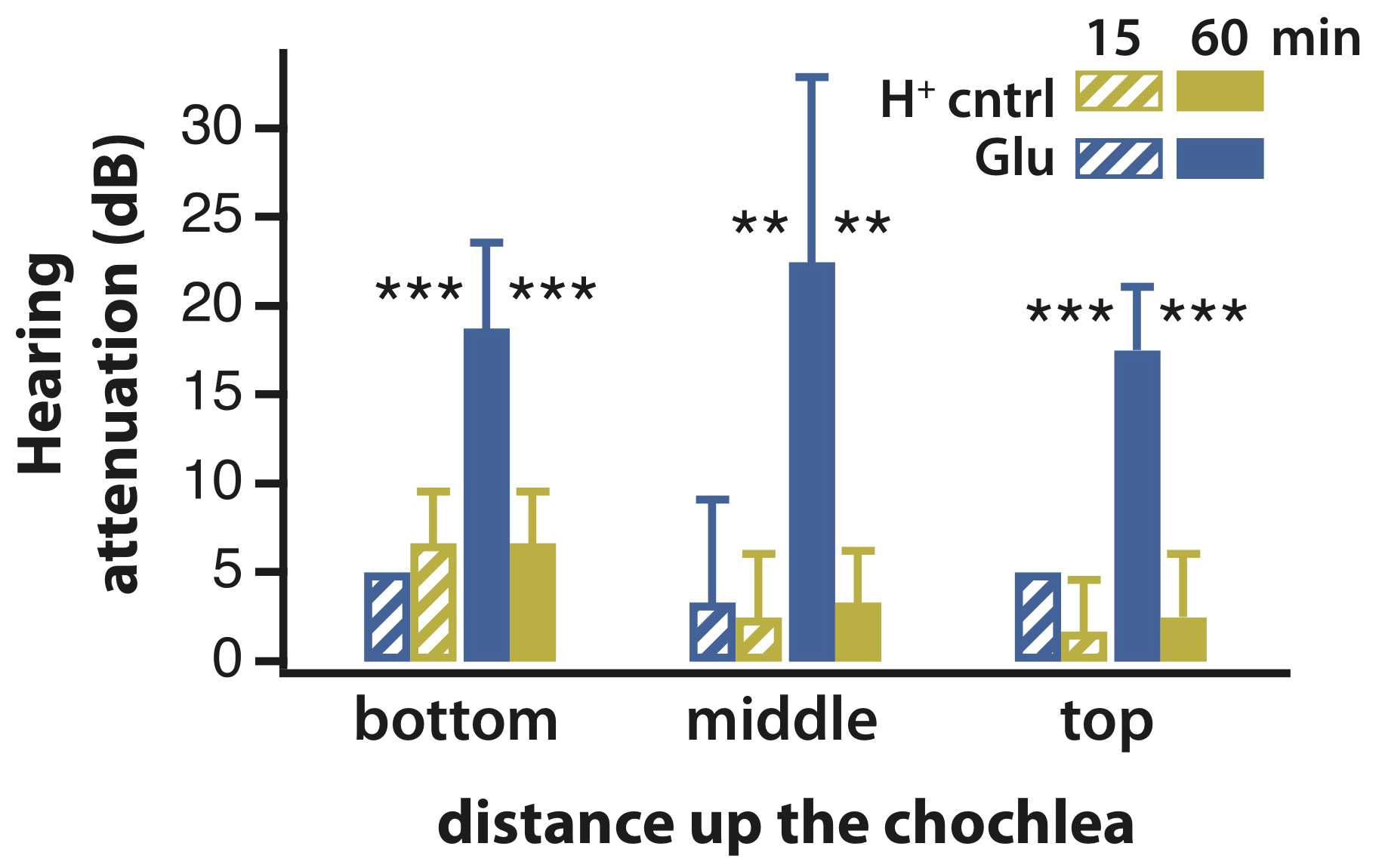
The in vivo OEIP was mounted for diffusive delivery into guinea pig cochleae, and the effect of glutamate was recorded in real time. After 60 min of delivery, significant hearing attenuation was observed, and histology indicated that only the inner hair cells were effected, demonstrating both efficacy and specificity. These experiments represented the first use of an organic bioelectronic device to control sensory function in a living mammal.
References
Products
The ion pumps have a the benefit of temporal control of the amount of ions delivered without flow of liquid or adding pressure to the system.
Flat ion pumps

The flat ion pump has the delivery point under the cell culture. In order to create a chamber for the cell cultures rings can be glued to the ion pumps. With the manufacturing methods applicable for the flat ion pump it is possible, in a simple way, to change the design desired by the user. The design of the device can be made to fit into different microscope setups. Typical applications is in vitro experiments. (Screen printed Flat ion pumps in the picture)
OBOETTE (in vivo)

The OBOETTE is a pipette that can deliver ions with temporal control of the amount of ions delivered without flow of liquid. The chambers of the double lumen OBOETTE is filled with the ions to be delivered. The ions are delivered at the tip, that for the moment has a diameter of 700μm. With the OBOETTE it is possible to deliver ions in both in vitro and in vivo experiments.(The OBOETTE with a tip
diameter of 700μm in the picture)